Anion Reactivity
Reactiviy of metal oxide anions
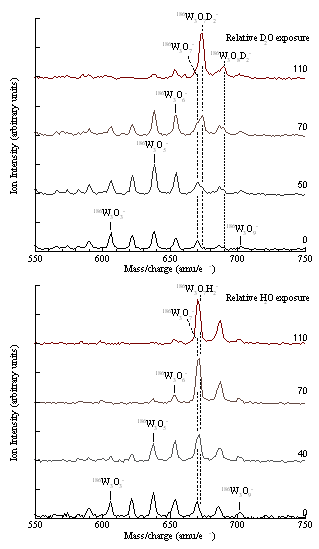 Photocatalytic decomposition of water- We have observed evidence of H2 evolution resulting from reactions between water and group 6 transition metal suboxide clusters.
Photocatalytic decomposition of water- We have observed evidence of H2 evolution resulting from reactions between water and group 6 transition metal suboxide clusters.
In the case of W3Oy− clusters, the sequential cluster oxidation/H2 reduction reaction terminates at W3O6− + H2O → W3O7H2−. To complete the catalytic cycle, release of OH or O2 from the cluster is necessary. We plan to use our OPO tunable light (410-709 nm) source to explore dissociative anion states that will regenerate the suboxides with photon energies overlapping with the solar spectrum.
Temperature Studies We have also begun the design of a variable temperature ion source (~298K – 1173K). This source will allow us to explore reactant barrier heights.


 The College of Arts
The College of Arts